INTRODUCTION
While it is well-established that dual antiplatelet (AP) therapy is more effective than aspirin alone in patients presenting with acute coronary syndromes (ACS), in recent times we have become a little more spoiled for choice when it comes to deciding just what that second AP agent should be. Traditionally we’ve reached for the closest bottle of clopidogrel, but in a situation not dissimilar to that seen with the veritable smorgasbord of oral anticoagulants now licensed for use, there are some “new” kids on the block worth taking into consideration.
In the beginning….. there was clopidogrel, loaded at 300mg early in ACS. Then came a 25,383 patient meta-analysis suggesting that 600mg was better than 300mg in STEMI going for PCI, so we started doing that. And lo, it was very good. Well, hey, it provided a statistically and clinically significant reduction in the incidence of MACE at 30 days, with no increase in bleeding, so one can’t really complain.
And on the second day… Daiichi Sankyo Company created prasugrel, (then got Ube Industries to manufacture it, and eventually Eli Lilly to sell it to the western world, because outsourcing is the way of the 21st century) and a couple of years ago we moved to prescribing it in a 60mg loading dose in STEMI (or any ACS that’s going to go for PCI, but we’re usually only really sure about the STEMIs that early in the game), but not to anyone over 75, or who’s had a stroke before, or who weighs less than 60kg, as these folks tend to bleed a little too easily.
Thus, all was well with the world, and the cardiologists were very pleased. But of course, the inevitable schism occurred… some heart specialists grew ever fonder of prasugrel, some felt that administration of clopidogrel was the one true way, and yet others (possibly mostly those with shares in AstraZeneca) clung steadfastly to their decision to use only ticagrelor, forsaking all other agents. Wracked with the anguish of conflicting opinions and requests from above, the Emergency Physicians temporarily chose to withdraw from the turmoil of indecision, shipping punters to the cath lab loaded only with aspirin and heparin.
So, what to make of this myriad selection of therapeutic options… this plethora of platelet-paralysing possibilities? One wonders… one wonders… And so we turn (as we should) to the evidence (such as it is) in an attempt to tease from the maelstrom a clear winner (if there is one) or possibly to realise that perhaps we might as well just toss a three-sided coin* after all…
*Those with a better than average grasp of the geometry of Platonic solids will likely prefer to toss a normal coin twice, or a tetrahedron once, ignoring one side, but needless pedantic accuracy just destroys the poetry of the moment, you know? Oh, wait…
PHARMACOLOGY (Stuff that sounds good on a lab bench)
CLOPIDOGREL (Plavix, Iscover)
- Irreversibly binds & inhibits the P2Y12 ADP receptor on platelets
- Is a pro-drug
- CYP-2C19 & CYP-3A4 are the most important pathways
- On average ~15% of the administered dose is processed to the active metabolite
- Wide, enormous, huuuuuge variation in how completely & quickly different people convert the pro-drug to the active metabolite
- Essentially a normal distribution between 0% and 100% platelet inhibition in the population
- 30% of people have an “inadequate” response (ie. No additional benefit over aspirin)
- Typically achieve ~60% platelet inhibition at around 6-8 hours after administration
PRASUGREL (Effient)
- Irreversibly binds & inhibits the P2Y12 ADP receptor on platelets
- Is a pro-drug
- CYP-3A4 is the most important pathway
- Rapidly & completely converted to the active metabolite
- So fast that 15 mins after administration, zero drug detectable in plasma
- Peak plasma concentration is reached within 30 minutes of administration
- Minimal variation in response between different people
- Causes faster, more complete and more consistent/reliable platelet inhibition than clopidogrel, achieving ~80% inhibition at 30 mins
- Patients known to be poor responders to clopidogrel, respond adequately to prasugrel
TICAGRELOR (Brilinta)
- Reversibly binds & inhibits the P2Y12 receptor on platelets
- Is an active drug right out of the box, directly binding P2Y12
- Faster than clopidogrel, but slower than prasugrel, maximum effect ~2 hours
- More complete platelet inhibition than either clopidogrel or prasugrel, at ~90%
- Causes coronary vasodilation (ticragelor is essentially adenosine with a couple of extra bits stuck on; in vivo it exerts adenosine-like effects)
- ~40% is converted to a metabolite with the same activity as the original drug
CLINICAL ENDPOINTS (Stuff we actually care about)
CLOPIDOGREL
This has become essentially the gold standard of care against which the other AP agents are judged. We know that adding clopidogrel to aspirin alone has benefits (risk of cardiovascular death/MI/stroke 9.3% vs 11.4% with aspirin alone), though these benefits clearly accrue well after the patient is out of the Emergency Department, as the drug is not doing anything useful to your platelets for at least 6 hours, and even then, with its incredibly variable efficacy it may not be doing terribly much for you, anyway.
We have already made mention of the evidence that 600mg tends to be better at preventing MACE at 30 days than 300mg is, without any additional bleeding risk. Looking a little further down the patient journey, there is evidence that 7 days of double-dose (150mg daily) clopidogrel, while of no additional benefit in medically managed ACS patients, does in fact reduce stent thrombosis and re-occlusion by 30% in those going for PCI. Doubling the dose does increase the bleeding risk, but not by much: 2.5% vs 2.0% for standard dosing.
One way of obviating the dilemma of unpredictable response to clopidogrel is to perform platelet function studies in the laboratory, and titrate the individual patient’s dose accordingly. This has been done using the vasodilator-stimulated phosphoprotein index (VASP to its friends). Using VASP to tailor doses, stent re-occlusion at 30 days was shown to fall to 0.5% versus 4.2% for the group receiving one-size-fits-none standard dosing. It’s perhaps also worth noting that some patients were given up to 2,400mg of clopidogrel (yes, really) based on their VASP, and 8% of those who were thus super-dosed still failed to respond adequately to the drug.
PRASUGREL
Is it any more effective than clopidogrel? Oh. Hell. Yes. In fact, I’m so excited about it I’m going to make a list:
- 19% reduction in (cardiovascular death / MI / stroke)
- 52% reduction in stent thrombosis
- 34% reduction in emergent re-do angio of the same vessel for occlusion
- 24% reduction in MI
Unfortunately, since prasugrel achieves these impressive results by more rapidly and effectively inhibiting platelet function than does clopidogrel, it also tends to cause more bleeding, to the tune of a rate of major bleeding of 2.4% vs 1.8% for clopidogrel. This is particularly problematic for patients who turn out to need urgent CABG, as they have a 5-fold higher risk of major bleeding during surgery, compared to patients on clopidogrel. This is a Bad Thing™. For those not requiring CABG, however, the vast majority of excess bleeding occurred in those over 75 years of age, those who had suffered a prior stroke (of any variety), and those weighing less than 60kg. When such patients are excluded, there is no significant difference in bleeding in non-CABG patients treated with prasugrel compared to clopidogrel. The perceptive reader may note that the standard contraindications to the administration of prasugrel in ACS are: age > 75yrs, history of prior CVA, or weight < 60kg. This is not a coincidence.
So, prasugrel certainly works much faster, more consistently, and more effectively than clopidogrel, but at the cost of a small increase in significant bleeding in PCI patients, and an enormous increase in the risk of bleeding for those unlucky enough to need an emergency CABG. In this latter group of patients, there is no easy solution, either, if you discover at angiography that the patient who was given prasugrel in ED now needs a CABG; the drug irreversibly binds P2Y12 and so, as with aspirin and clopidogrel, you have to wait for your affected platelets to die of natural attrition and be replaced before you recover normal platelet function (7-10 days). For this reason, some clinicians strongly advocate that prasugrel should be withheld until the coronary artery anatomy and lesions have been defined at angiography, so the drug can be avoided in those with disease requiring CABG rather than stenting.
TICAGRELOR
What about this one… is it a contender? Similar to prasugrel it confers significant improvements in efficacy over clopidogrel:
- 16% reduction in (cardiovascular death / MI / stroke)
- 23% reduction in stent thrombosis
- 21% reduction in cardiovascular death
- 22% reduction in mortality
In all-comers given ticagrelor before anyone has figured out if you need a CABG or not, there is no difference in significant bleeding between patients on ticagrelor or clopidogrel. If you isolate those not getting a CABG, however, there is a small increase in bleeding: 4.5% vs 3.8% with clopidogrel.
This somewhat counter-intuitively favourable bleeding profile in CABG patients is almost certainly largely due to the fact that this drug’s effects are fully reversible. When the drug wears off, your existing platelets are functional again. Thus if you can be medically managed for 24 hours before you get your CABG, then your bleeding risk is, predictably, quite a bit lower than if your platelets were definitively stuffed for the next week or more by clopidogrel or prasugrel.
SUMMARY
The table below summarises the salient characteristics of each drug. The composite endpoint is (cardiovascular death / MI / stroke), and the other efficacy column denotes utility in preventing stent thrombosis.
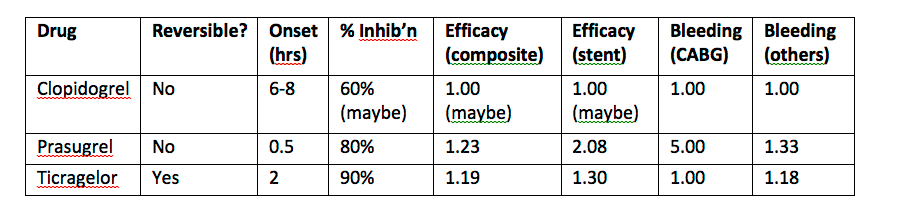
Note: Efficacy figures are derived from the relative risk reduction / increase and standardised to units where clopidogrel’s value is a baseline of 1 unit. Bleeding risk is simply a hazard ratio with the risk of bleeding on clopidogrel expressed as 1 unit.
DISCUSSION
Prasugrel and ticagrelor both offer significant improvements in efficacy, and especially in reliability, over clopidogrel. They both come with an increased risk of bleeding, though the absolute risk increase is quite small at 0.6% and 0.7% respectively. In my opinion this increased risk is clearly outweighed by the reduction in cardiovascular morbidity and mortality conferred by these two drugs. Ticagrelor, particularly, offers a 22% RRR in death (pretty solid endpoint, that one) at the cost of an 18% relative increase in risk of bleeding (that probably won’t kill you, and indeed the bleeders were included in the analysis that yielded the overall increased mortality figure in the first place).
Prasugrel would be the poster child here, were it not for its tendency to make CABG patients bleed +/- die on the table at around 500% the rate that clopidogrel or ticagrelor do. One could argue only around 5% of STEACS patients presenting to hospital will be unstentable and need urgent CABG, and if you crunch the numbers, giving prasugrel to all-comers will still save lives overall, at the expense of killing a handful of CABG-dependent patients.
In a less utilitarian world, however, where we do actually care what happens to individual patients, not just the herd, I would submit that ticagrelor achieves an optimal middle ground when it comes to early therapy, before you know if they’re going for a CABG. It’s faster and more reliable than clopidogrel, with only marginally increased risk of bleeding, and you can turn it off almost instantly if the patient requires a CABG.
RECOMMENDATIONS
I see two sensible options here:
OPTION 1 – Give no second-line AP agent in ED
- Give aspirin 300mg PO
- Give heparin 5,000 units IV
- Expedite journey to cath lab
- Once the cardiologist knows they can stent them, they give prasugrel 60mg
OPTION 2 – Give ticagrelor in ED
- Give aspirin 300mg PO
- Give heparin 5,000 units IV
- Give ticagrelor 180mg PO
- Expedite journey to cath lab
- Whether they need a CABG or not is irrelevant
If we assume a worst case scenario of 90 mins between when we might give a drug (or not) and when the cardiologist obtains angiographic images enabling them to determine if the patient needs an urgent CABG, then the ticagrelor (2 hours onset) we give downstairs, or the prasugrel (30 mins onset) they give upstairs, will be reaching peak effect at approximately the same time.
If the cath lab is faster than that (and they usually are), then the wait/angio/prasugrel approach will achieve platelet inhibition earlier than giving ticagrelor in ED, and for the PCI population, prasugrel offers slightly better efficacy than ticagrelor, albeit at the cost of non-reversibility should bleeding complications ensue.
Sadly, no-one has done the study to tell us if giving early ticagrelor PLUS prasugrel a bit later once you know there’s no CABG on the horizon, carries any greater bleeding risk than either drug alone. If the answer is “no”, then we should go with OPTION 2, give everyone ticagrelor, and then the cardiologists can add prasugrel if they wish. But we just don’t have the data.
THE ABSOLUTE NITTY GRITTY BIT
We should give ticagrelor 180mg PO, or nothing.
REFERENCES
Note that while claims made above are sourced from various pieces of primary research literature (PLATO, TRITON-TIMI, etc.) the following single review article covers things extremely well and the bibliography contained therein is sufficient to direct you to the relevant individual studies:
http://www.ncbi.nlm.nih.gov/pubmed/23933898
It should be available online for free, but I have the full-text PDF if anyone would like it emailed to them.


